Title: NASH Surprises and Interpretation of Clinical Trials
Author: Peter G. Traber, MD
Introduction
2019 will usher in a new chapter in NASH drug development with data reported on the first phase 3 clinical trials. Gilead Bioscience’s studies of selonsertib, STELLAR 3 in NASH with stage 3 fibrosis and STELLAR4 in NASH cirrhosis, will report results in first half of 2019. Intercept Pharmaceutical’s study of obeticholic, REGENERATE in NASH with stage 2-3 and high-risk stage 1 fibrosis, will report results in the first quarter, and Genfit’s study of elafibranor, RESOLVE-IT in stage 1-3 fibrosis, will report results in the fourth quarter. These studies will set the bar for the first wave of NASH therapeutics.
What can we expect in improvement of NASH is dependent on the natural history of NASH without therapy. Of course, each of these trials has a placebo control group that will provide that information in the specific groups of patients studied, but what do we know now about the natural history? In fact, we know surprisingly little and several recent publications provide some startling new insights.
Fibrosis is key determinant of mortality, with a twist
While it is clear that the stage of fibrosis is the most important determinant of adverse clinical outcomes in patients with NASH, a recent longitudinal, observational study adds critical new information (Gastroenterology 2018;155:443-457).
Mortality in this study was very low over 10 years in NASH patients with advanced, stage 3 fibrosis (Figure 1), and the most important clinical events were cardiovascular and non-hepatic cancers (Figure 2). Mortality was much higher in patients with compensated cirrhosis, with an important twist. Patients classified as Child class A with a score of 6 had a much worse prognosis than patients with a score of 5, including higher rates of death/transplant, decompensation events, and hepatocellular carcinoma (Figure 1 and 2).
Figure 1. Adjusted overall survival without transplant according to fibrosis stage and Child Turcotte Pugh (CTP) score
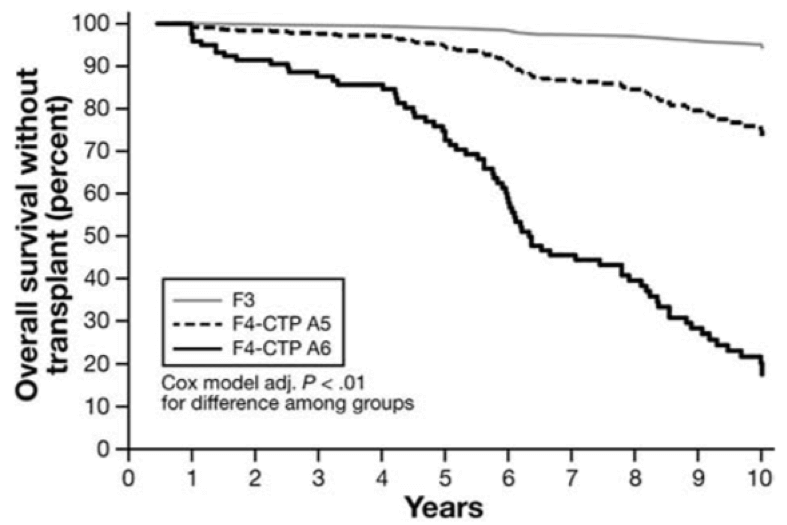
Figure 2.
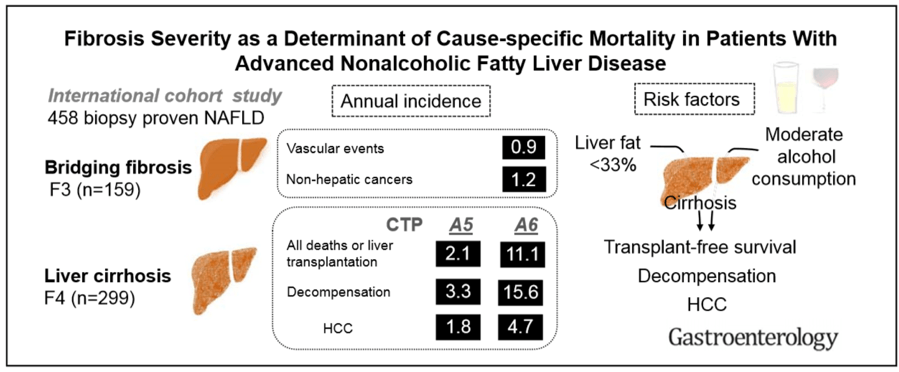
These findings may be critically important for interpretation of NASH cirrhosis trials, because none of the ongoing trials to my knowledge, stratify for CTP score within Child classes. Therefore, any analysis of such subsets will be post-hoc, a problem in phase 3 clinical trials. These data suggest that Childs class A patients with compensated NASH cirrhosis will be a heterogenous population regarding clinical outcomes. Additionally, some studies may extend entry criteria to include patients with a CTP score of 7, which is just into the Child Class B group of patients, complicating the situation even more.
NASH Fibrosis and Cirrhosis Trials Show Surprising Placebo Results
Recently, two large, long-term treatment trials of simtuzumab were reported (Gastroenterology;155:1140-1153), one in NASH with stage 3 fibrosis and the other in NASH cirrhosis. Unfortunately, the putative anti-fibrotic simtuzumab had no effect on the progress of the disease, but analysis of the placebo groups yielded a wealth of surprising information.
The simtuzumab NASH stage 3 fibrosis study was a large, randomized controlled clinical trial (n=219) evaluating two doses of simtuzumab after 48 and 96 weeks of therapy. The placebo group actually showed a decrease in liver collagen on biopsy over the 96-week trial (Figure 3). There was no difference between placebo and treatment groups in mean change in hepatic collagen by morphometry, the primary endpoint.
Figure 3. Simtuzumab in NASH patients with bridging fibrosis
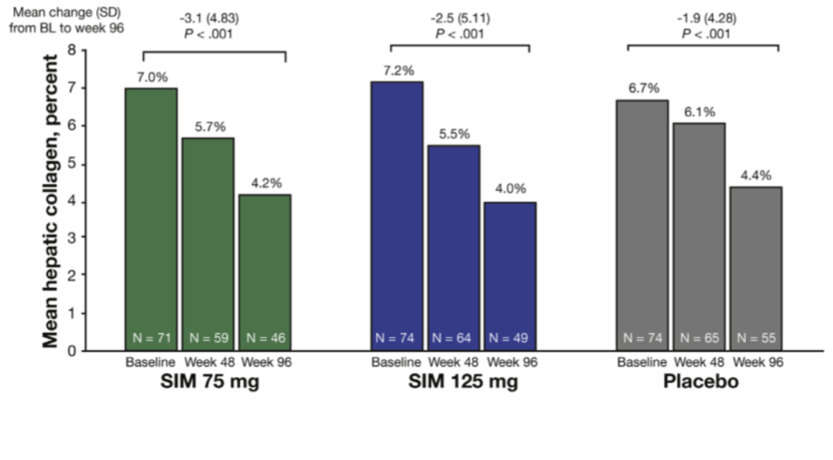
In addition, 23% placebo patients had at least a one stage improvement in NASH-CRN score, and there was no difference in SIM groups. Finally, 20% of placebo patients progressed to cirrhosis over mean observation of 30 months. The same percentage of placebo patients improved as progressed to cirrhosis, demonstrating the heterogeneity of this population. The paper discusses potential reasons for these observations.
The simtuzumab NASH cirrhosis study was a large, randomized controlled clinical trial (n=258) evaluating two doses of SIM after 48 and 96 weeks of therapy. The primary endpoint was change in HVPG (hepatic venous pressure gradient) at week 96 and 67% of patients had clinically significant portal hypertension (> 10 mmHg) and 43% of patients had esophageal varices.
There was no difference between placebo and treatment groups and, although it was not statistically significant, there was a trend towards improvement of HVPG over the 96 weeks. In placebo patients with a median observation of 29 months, 8% had at least a one stage improvement in NASH-CRN fibrosis score, and there was no difference in the simtuzumab groups. Therefore, the trend towards improvement over 96 weeks was like the NASH stage 3 patients.
Summary
At the outset of clinical development programs in NASH with advanced fibrosis and cirrhosis, most would have certainly anticipated that placebo patients would get worse over 96 weeks. Why they got better can be speculated on, but the important issue this natural history should be anticipated as phase 3 trials report data in 2019. Additionally, this will be important to take into consideration in designing new clinical trials and choosing patient populations to evaluate. It is an exciting time as we learn more about the natural history of this enigmatic disease.
Additional NASH Insight
- NASH Cirrhosis Space Poised to Heat Up
- Is Rapid Reversal of Liver Fibrosis Possible?
- Industry Strategies for NASH Combination Therapies
- Agencies Clarify NASH Endpoints — But Don’t Harmonize
For information on my consulting work please click here.

