Types of cellular immunotherapy:
Cellular immunotherapies involve modifying autologous or allogeneic immune cells through genetic engineering or modulation of the protein expression profile, then administering these modified cells to the patient1. In 2010, the first autologous dendritic cell (DC) vaccine sipuleucel-T (Provenge; Dendreon Pharma), was approved by the FDA for the treatment of asymptomatic metastatic stage IV prostate cancer. This was followed in August 2017 by the landmark approval of the first CAR-T cell therapy, tisagenlecleucel (Kymriah; Novartis), which was followed swiftly by the approval of axicabtagene ciloleucel (Yescarta; Kite Pharma) in October 2017. Since then, the cellular immunotherapy space has grown exceptionally fast, with biotech and pharmaceutical companies developing several different types of cellular immunotherapies in a race to take the biggest market share in the numerous oncology (and other) indications that present a huge unmet need.
The are eight main types of cellular immunotherapy:
- Chimeric antigen receptor (CAR)-T cell therapy is a huge advancement in personalized cancer treatment. It involves genetically modifying a patient’s own cells (autologous) or a donor’s cells T cells (allogeneic) to express a CAR, expanding these CAR-T cells ex-vivo and then infusing them into the patient’s body where they can attack and destroy cancer cells.
- T cell receptor (TCR) therapy is an adoptive cellular therapy (ACT) that has arguably shown more success in targeting solid tumors than CAR-T therapies 2 and have a wider range of targets than CAR-T therapies. However, TCRs are major histocompatibility complex (MHC) restricted and different versions of each product have to be made for each patient haplotype 3.
- Natural killer (NK) cells can be modified into CAR-NK therapies and used to target malignant cells, producing short-lived rapid responses against them. They also have some advantages over CAR-T therapies mostly in terms of safety, as they do not require strict human leukocyte antigen (HLA) matching and do not carry the risk of graft-versus-host disease (GvHD), making them suitable candidates for developing allogeneic therapies. Furthermore, they come without the risk of developing cytokine release syndrome (CRS) due to their short-lived nature 4.
- Gamma-delta T cells (γδ-T cells) are on the junction of the innate and adaptive immune systems. They are another immune cell, alongside alpha beta (αβ) T lymphocytes and B lymphocytes, that can be used to develop cellular immunotherapies. γδ-T cells possess similar cytotoxic features as αβ T cells however, they offer other innate-like features including expressing certain NK cell receptors (NCR). Furthermore, they have the unique ability to recognize multiple antigens in an MHC-unrestricted fashion and thus also do not trigger GvHD. They undergo maturity in the thymus while maintaining a preactivated state, thereby not requiring clonal expansion or differentiation into an effector T cell phenotype when activated 5. Interestingly, studies have suggested that γδ-T cell infiltration correlates higher with survival than any other immune cell 6.
- ACT with tumor-infiltrating lymphocytes (TIL) predates CAR-T therapies and has been an anti-cancer treatment of interest for the last decade 7. In trials, the approach has shown a lot of promise in the treatment of melanoma, cervical cancer 8 and more recently for the treatment of non-small cell lung carcinoma (NSCLC) 9.
- Cytokine induced killer (CIK) cells are a subset of polyclonal T-effector cells possessing both NK and T cell properties. They possess MHC-unrestricted cytotoxicity against multiple different tumor target cells and preclinical studies have suggested that they have strong anti-tumor effects against both blood cancers and solid tumors. A big advantage of CIK cells is that they can be expanded to very large numbers ex-vivo. 10.
- Macrophages are cells of the innate immune system that act as both phagocytes and antigen-presenting cells (APC). CAR-macrophages can be developed as cancer immunotherapies and may in fact be better able to target solid tumors than traditional CAR-T therapies, as they can modulate the tumor microenvironment 11.
- Dendritic Cells (DC) play a crucial role in immuno-surveillance. They are present throughout the body and monitor their surroundings for antigens. If stimulated, they undergo maturation, migrate to the lymphoid organs and activate T cells and B cells 12. They are also the most potent APC and can proficiently perform antigen cross-presentation, presenting antigens to both CD4+ and CD8+ T cells. This makes DCs potentially useful as vaccine platforms for inducing anti-tumor cytotoxic T lymphocyte (CTL) immune responses 13. While the first approved DC vaccine was autologous, allogeneic DC vaccines made from donor DCs are also being developed 14.
The Landscape
Technology Type:
CD-19 targeted CAR T cells, of which Kymriah and Yescarta have been approved, demonstrated before unseen levels of clinical efficacy in acute lymphoblastic leukemia (ALL) patients who had exhausted all available treatment options. Kymriah was first approved for pediatric ALL, but the label was then expanded to treatment of the most common form of non-Hodgkin’s lymphoma (NHL) in adults. Yescarta was approved for treating several types of adult NHL 15. Both therapies come with high price tags ($475,000 to treat ALL and $373,000 to treat NHL with Kymriah; $373,000 to treat NHL with Yescarta) 16 and large-scale manufacturing issues that played a role in preventing wider adoption of both therapies 17. Nevertheless, interest in CAR-T therapy development continues to prevail.
Since approval in 2010, Provenge has failed to live up to expectations and has shown limited success in producing significant objective clinical responses 12. This seems to have been a common problem with other DC vaccines and is mostly why no other pure DC vaccine has since been approved by the FDA. Other types of DC vaccines are approved for use in some countries e.g. CreaVax-RCC, an autologous DC vaccine for treating metastatic renal cell carcinoma (RCC) in South Korea 18. Hybricell (Genoa Biotecnologia) is a DC-tumor cell hybrid vaccine approved in Brazil for treating melanoma and RCC. It has been shown to prevent disease progression in 80% of melanoma or kidney tumor patients 19. Finally, DCVax-L 20 is an autologous DC vaccine that was granted hospital exemption (lasting five years) for treatment of glioblastoma (GBM) in Germany since 2014 21. Although the company and the associated clinical data have been the subject of significant controversy 22.
The current global development pipeline consists of hundreds of cellular immunotherapies being pushed forward to address the huge unmet need in a range of different cancers (Figure 1). Many more are autologous than allogeneic. Across the spectrum of cellular immunotherapy types, there are just as many programs at preclinical stage as there are at clinical stage, but this is not true for DC vaccines for which there are 87 in clinical development and only 21 in preclinical development. The National Cancer Institute (NCI) is developing the most DC vaccines with seven in the pipeline, and has a whole research branch dedicated to the development of cancer vaccines 23.
CAR-T therapies are the most highly represented, with 592 in development and two approved (Figure 1). The popularity of CAR-T cell therapies has been driven by their spectacular clinical success in blood cancers 24. Innovative Cellular Therapeutics is most active in the CAR-T therapy space with 15 therapies in development (Figure 2). The company is using its next generation CAR-T technology to build a portfolio of therapies, targeting a wide range of indications including both solid and liquid tumors 25.
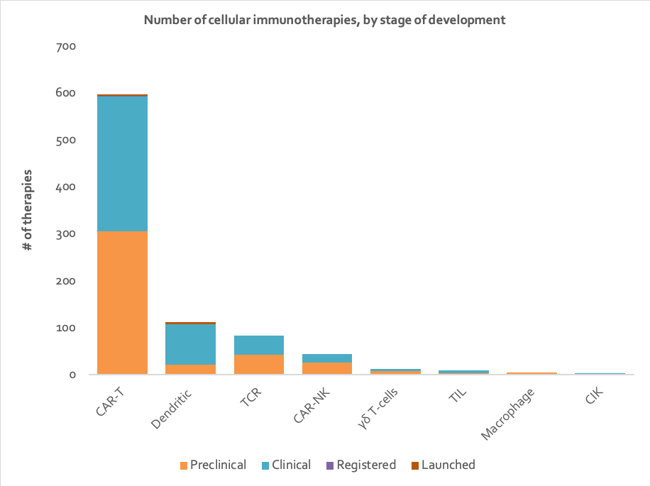
Figure 1: Stacked column chart depicting number of therapies in each stage of development for each type of cellular immunotherapy (data from Clarivate’s Cortellis - accessed H1 2020).
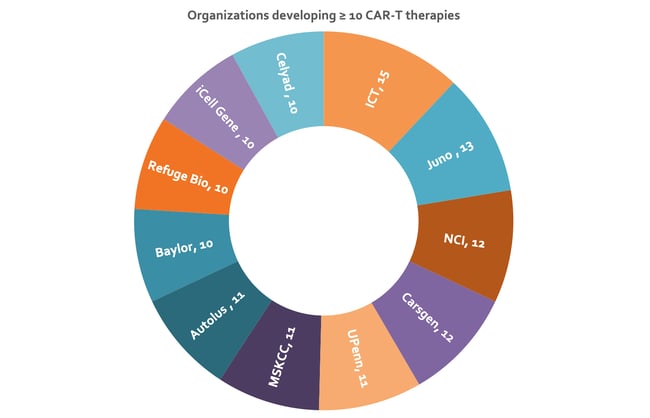
Figure 2: Sunburst chart depicting organizations with ≥ ten CAR-T therapies under development. Nearly 6% of the assets represented here are under collaboration, resulting in some duplication in counts of number of therapies (data from Clarivate’s Cortellis - accessed H1 2020). Baylor, Baylor College of Medicine; UPenn, University of Pennsylvania; MSKCC, Memorial Sloan Kettering Cancer Center; NCI, National Cancer Institute; ICT, Innovative Cellular Therapeutics.
Adaptimmune Therapeutics and Medigene seem to be the most active in TCR therapy development. Adaptimmune is considered a leader in the TCR field due to its innovative SPEAR technology 26. Medigene aims to develop several cancer-specific T cells for treating large tumor burdens 27.
NantKwest has developed the novel taNK platform to develop CAR-NK therapies against a wide range of both solid and liquid tumors 28; it is the most active company in the CAR-NK field with eight therapies in the pipeline.
There are several companies developing γδ-T cell therapies including Beijing Doing Biomedical, TC Biopharm, Adicet Bio, Takeda Pharmaceutical in collaboration with GammaDelta Therapeutics, and Incysus Therapeutics in collaboration with University of Alabama at Birmingham. In collaboration with Blue Rock Therapeutics, Editas Medicine is developing an allogenic γδ-T cell therapy that utilizes CRISPR/Cas9 gene editing. Cytomed Therapeutics is developing an off-the-shelf allogeneic NKGD2L targeted CAR- γδ-T cell therapy currently in a phase 1 clinical trial in subjects with relapsed or refractory solid tumors.
In the TIL therapy space, those developing autologous TIL therapies include Immetacyte and the University of Copenhagen. Cellular Biomedicine Group is developing a neoantigen activated TIL to be used in combination with an anti-programmed cell death-1 (PD-1) therapy. Iovance Biotherapeutics and The Fred Hutchinson Cancer Research Center are also in the TIL race, each with three therapies in development.
CIK therapies appear to mostly be in development in China with only four organizations leading the effort. Beijing Biohealthcare Biotechnology is developing a CIK therapy made up of autologous DCs co-cultured with CIK cells 29, No 307 Hospital of the Chinese People’s Liberation Army is developing a CIK therapy that is used in combination with a DC vaccine 30, Shenzhen Hornetcorn Bio-technology Company is developing a CIK therapy which uses pembrolizumab-activated autologous D-CIK cells 31, and People’s Hospital Peking University is developing a CIK therapy that utilizes DCs loaded with 6B11minibody (a vaccine against human ovarian cancer) that are co-cultured with peripheral blood mononucleocytes (PBMNC) and stimulated by various cytokines, to form 6B11mini-ovarian-CIK cells 32.
The University of California San Diego is developing a macrophage therapy that utilizes iSNAPS, which are integrated ‘sensing and activating’ proteins that have been inserted into macrophages, thereby enhancing the ability of the macrophages to engulf and destroy cancer cells 33. Other companies in this space include Carisma Therapeutics which is developing novel CAR-macrophage therapies. The technology uses CARs to provide target recognition to human macrophages, which in turn triggers macrophage phagocytic and antigen-presenting functions to attack cancer cells.
Targets
Target selection has a big impact on the efficacy of a therapy. For example, in order to create a CAR-T therapy that successfully targets solid tumors, it is wise to choose well-known tumor targets specifically within solid tumors. Examples of such targets include EGFR which is found in liver, colorectal and gastric cancers; or GD2 which is found in gliomas and neuroblastomas. But one issue complicating targeted cellular immunotherapies is tumor escape via tumor plasticity, which ultimately leads to target loss. Targeting multiple tumor antigens can help to deal with this. As such, cellular immunotherapies that are multi-targeting or bispecific are increasingly favored. Choosing a tumor target with high specificity is also crucial, as low specificity can lead to on-target off-organ toxicity - this ultimately means researchers need to identify tumor targets that are not widely found on normal functioning cells 34. Some therapies target tumor associated antigens (TAA) which are present in normal tissues but at lower levels than on tumors; this carries an inherent potential risk of side effects 35. All of this makes tumor specific antigens (TSA) such as neoantigens an attractive alternative target approach, as these targets are only found in tumors 36. In conclusion, target selection for a cellular immunotherapy involves finely balancing therapy efficacy with target specificity and the risk of tumor escape. Competition also plays a big part in the selection process. Popular tumor targets such as CD19 and BCMA are not likely to be the primary target of choice for late-comers to this space, forcing researchers to look for unique and novel tumor targets 37.
CAR-T Therapies
CD19 is the tumor target most popular amongst CAR-T therapies, with a staggering 116 programs (see Figure 3). This includes the two launched CAR-T therapies, (Kymriah and Yescarta). CD19 has traditionally been considered an ideal target for CAR-T therapies as it is a cell surface molecule that is expressed on most B cell malignancies, including chronic lymphocytic leukemia, acute ALL, and NHL. Furthermore, since CD19 is not expressed on hematopoietic stem cells (HSC), elimination of all CD19+ B cells in the body is clinically manageable using intravenous immunoglobulin (IVIG) 38.
BCMA is the second most common tumor target of CAR-T therapies, with 42 anti-BCMA programs. This is likely because BCMA is the most promising tumor target for treating multiple myeloma (MM), an as yet incurable disease due to its complex genetic heterogeneity and high relapse rate after treatment 39.
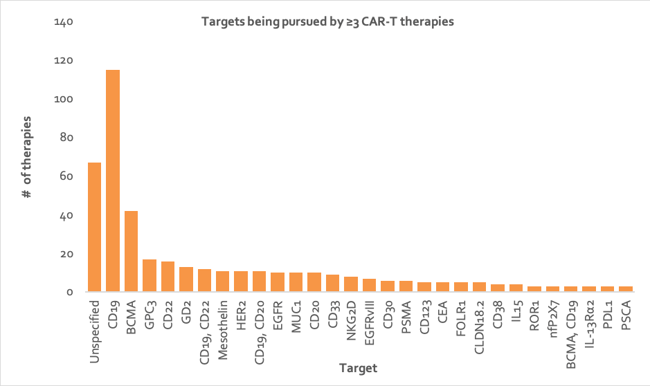
Figure 3: Column chart depicting number of CAR-T cell therapies directed at each tumor target, where the target or target combination is being pursued by at least three CAR-T cell therapies (data from Clarivate’s Cortellis - accessed H1 2020). CD, cluster of differentiation; BCMA, B-cell maturation antigen; GD2, disialoganglioside; GPC3, glypican-3; HER2, human epidermal growth factor receptor 2; EGFR, epidermal growth factor receptor; MUC1, mucin 1; CEA, carcinoembryonic antigen; EGFRVIII, epidermal growth factor receptor variant III; PSMA, prostate-specific membrane antigen; ROR1, receptor tyrosine kinase like orphan receptor 1; IL15, interleukin-15; nfP2X7, non-pore functional P2X7; FOLR1, folate receptor 1; CLDN18.2, claudin-18.2; IL-12Rα2, interleukin 12 receptor alpha 2; NKG2D, natural-killer G2D; PDL1, programmed death-ligand 1; PSCA, prostate stem cell antigen.
TCR Therapies
NYESO1 is the most popular tumor target for TCR therapies, with 16 programs (Figure 4). It is the most promising cancer testis antigen (CTA) target to date. Its tumor expression is distinctly associated with activation of an immune response, making it highly suitable for immune-based interventions in a wide range of cancers 40.
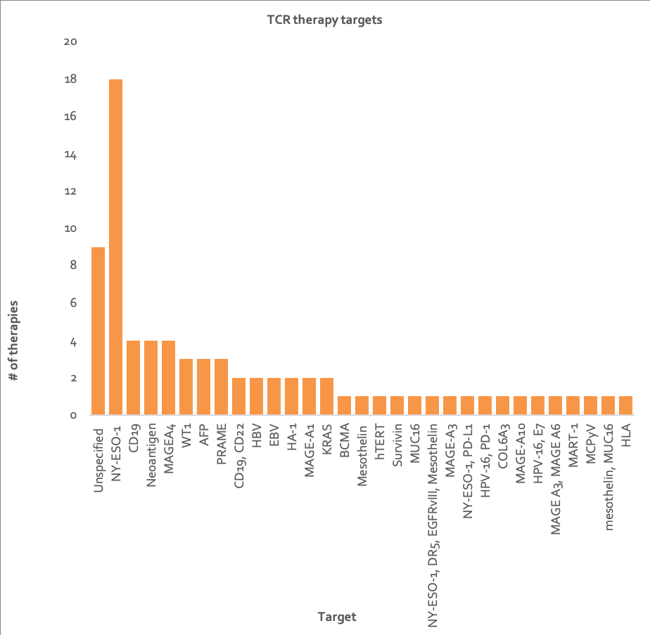
Figure 4: Column chart depicting number of TCR therapies directed at each tumor target or target combination (data from Clarivate’s Cortellis - accessed H1 2020). HLA, human leukocyte antigen; MUC16, mucin 16; MCPγV, merkel cell polyomavirus; PRAME, melanoma antigen preferentially expressed in tumors; MART-1, melanoma antigen recognized by T cells 1; MAGE, melanoma antigen; HPV, human papillomavirus; COL6A3, collagen alpha-3(VI); HA-1, hemagglutinin 1; PD-1, programmed cell death protein 1; NY-ESO, New York esophageal squamous cell carcinoma; PD-L1, programmed death-ligand 1; EBV, epstein-barr virus; DR5, death receptor 5; EGFRvIII, epidermal growth factor receptor variant III; AFP, alpha-fetoprotein; hTERT, telomerase reverse transcriptase; WT1, wilms' tumor protein 1; CD, cluster of differentiation; BCMA, B-cell maturation antigen.
DC Vaccines and CAR-NK Therapies
Neoantigens are the most common target for DC vaccines, with over 30 under development and all four marketed DC vaccines (Sipeuleucel-T, DC-VAX-L, HybriCell and CreaVax-RCC) targeting neoantigens 19, 20, 40, 41. They are essentially a class of TSA that possess stronger immunogenicity and affinity towards MHC than TAAs 36. Furthermore, unlike TAAs which are also found on healthy tissues, neoantigens are only found on tumors so the patient’s immune system recognizes these antigens as non-self 42. CAR-NKs are being developed as a safer alternative to CAR-T therapies 4 and so CD19 is the most popular tumor target approach for CAR-NK therapies, for reasons similar to CAR-T therapies.
CIK, γδ-T Cell, Macrophage and TIL Therapies
Most targets of CIK therapies in development are unspecified and so it is not possible to draw any conclusions about which are the most promising. As mentioned above, Cytomed Therapeutics is developing a CAR-γδ-T cell therapy targeting NKGD2 ligands 43, and Adicet Bio has revealed that it is developing an anti-CD20 CAR-γδ-T cell therapy and an anti-GPC3 CAR-γδ-T cell therapy 44. Carisma Therapeutics’ CAR-macrophage therapy targets HER2 which has a long history in anti-cancer therapy. All TIL therapies are effectively bespoke, in that they use a patient’s tumor material and therefore target specific neoantigens expressed within the tumor 7.
Indications
Currently, CAR-T therapies are more successful in treating hematological neoplasms than solid tumors, as locating, entering and surviving the solid tumor environment is a significant technical hurdle CAR-T therapies have yet to overcome 45. In contrast, TCR therapies have been more successful in treating solid tumors as they can target a wider range of antigens (and especially those found on solid tumors) largely due to their ability to recognize intracellular proteins expressed as peptides on MHC class I molecules. In contrast, CAR-T therapies are limited to membrane-expressed antigens 46. Therapies targeting neoantigens have also shown a lot of success in treating solid tumors such as melanoma, which may be why the focus of the majority of DC vaccines and all TIL therapies seems to be solid tumors 47 (see Figure 5).
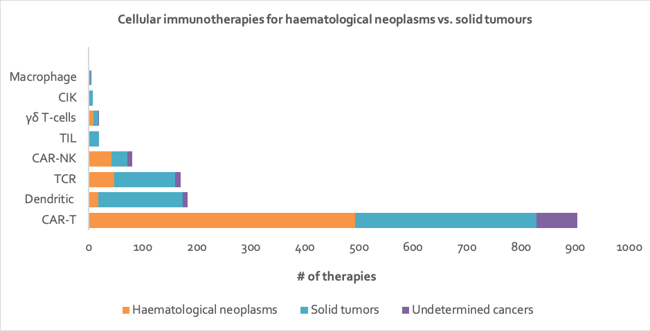
Figure 5: Stacked bar chart depicting number of cellular immunotherapies targeting hematological neoplasms vs. solid tumors, by technology type. Of all the therapies represented here, over 2% target multiple tumor types, resulting in some duplication in counts of therapies (data from Clarivate’s Cortellis - accessed H1 2020).
CAR-T Cell Therapies
Given that the most common target antigen for CAR-T therapy, CD19 is found on the surface of B-cells 48, it is no surprise that the indication being pursued by the most CAR-T therapies is ALL, with 97 programs (see Figure 6). ALL is also the most common type of leukemia in children 49 and presents a great unmet need. The second most popular indication being pursued by CAR-T therapies is MM, which correlates with BCMA being the second most common target of CAR-T therapies (see Figure 3) 39.

Figure 6: Waterfall chart depicting indications being pursued by at least five CAR-T therapies. Approximately 20% of the assets represented here are under collaboration, resulting in some duplication of counts of therapies (data from Clarivate’s Cortellis - accessed H1 2020). ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; B-ALL, B-cell ALL; CLL, chronic lymphocytic leukemia; CRC, colorectal cancer; DLBCL, diffuse large B-cell lymphoma; HCC, hepatocellular carcinoma; FCL, follicular lymphoma; MCL, mantle cell lymphoma; MM, multiple myeloma; NHL, non-hodgkin lymphoma; NSCLC, non-small cell lung carcinoma; RCC, renal cell carcinoma.
TCR Therapies
Ovarian cancer, NSCLC, melanoma, sarcoma and MM are targeted by the most TCR therapies (Figure 7). NYESO1-specific TCR-T cells, the most common target of TCRs, have undergone the most investigation for their therapeutic potential for synovial cell sarcoma, melanoma, and myeloma 2.
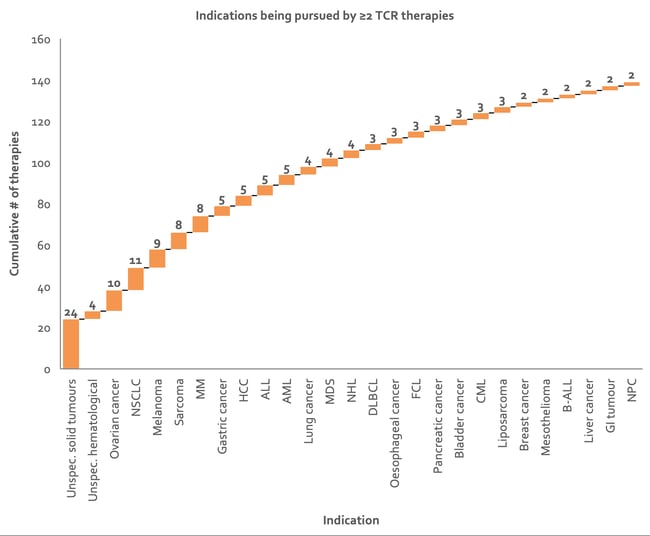
Figure 7: Waterfall chart depicting indications being targeted by at least two TCR therapies. Over 20% of the assets represented here are under collaboration, resulting in some duplication of counts of therapies (data from Clarivate’s Cortellis - accessed H1 2020). NSCLC, non-small cell lung carcinoma; MM, multiple myeloma; DLBCL, diffuse large B-cell lymphoma; HCC, hepatocellular carcinoma; FCL, follicle cell lymphoma; ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; MDS, myelodysplastic syndrome; B-ALL, B-cell ALL; NHL, non-hodgkin lymphoma; CML, chronic myeloid leukemia; NPC, nasopharyngeal cancer.
TIL Therapies
Melanoma is also a popular choice of indication for TIL therapies and has been well studied, consistently showing favorable results with response rates of over 50% and durable complete response rates (CRR) of 20% in patients who have not been successfully treated with other therapies, including checkpoint inhibitors (CPI) 50. TIL therapy success in melanoma may be somewhat linked to the high mutational load seen in this cancer type, which is typically associated with greater treatment benefit and greater clinical response 51. Iovance Biotherapeutics is currently running pivotal trials of its advanced cervical cancer and metastatic melanoma TIL therapy, aiming to obtain FDA approval in the second half of 2020 52.
CAR-NK therapies
The hematological cancers MM, AML, and B-cell lymphoma are currently the most common indications for CAR-NK therapy (Figure 8), which is in essence being developed as an allogeneic alternative to CAR-T therapy 4. MM cells are highly heterogeneous and the currently known surface markers are still unclear and so MM remains an incurable malignancy with a poor overall prognosis. The prognosis of AML in elderly subjects has also not yet been significantly improved with current therapy options.
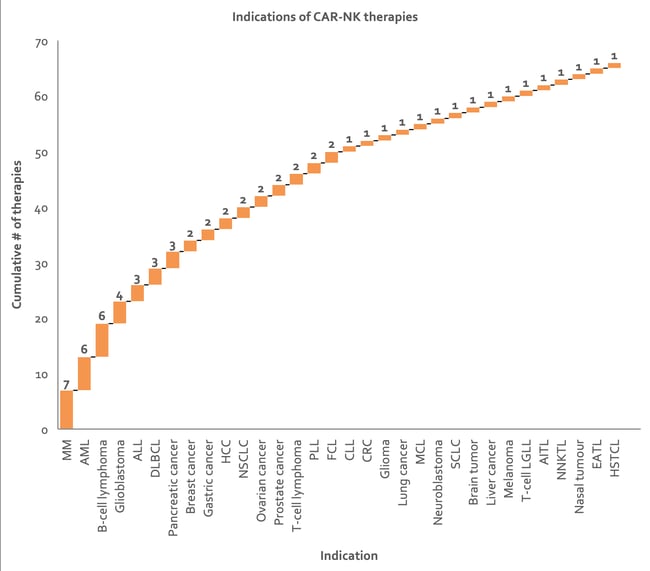
Figure 8: Waterfall chart depicting indications being targeted by CAR-NK therapies. Approximately 17% of the assets represented here are under collaboration, resulting in some duplication in counts of number of therapies (data from Clarivate’s Cortellis - accessed H1 2020). ALL, acute lymphoblastic leukemia; AML, acute myeloid Leukemia; CLL, chronic lymphocytic leukemia; CRC, colorectal cancer; DLBCL, diffuse large B-cell lymphoma; HCC, hepatocellular carcinoma; MCL, mantle cell lymphoma; NSCLC, non-small cell lung carcinoma; SCLC, small cell lung carcinoma; PLL, prolymphocytic leukemia; FCL, Follicle cell lymphoma; T-cell LGLL, T-cell large granular lymphocytic leukemia; AITL, angioimmunoblastic T-cell lymphoma; NNKTL, nasal natural killer (NK)/T-cell lymphoma; EATL, enteropathy associated T-cell lymphoma; HSTCL, hepatosplenic T-cell lymphoma.
DC Cell Vaccines
There are 21 DC vaccines targeting melanoma and 15 against NSCLC (see Figure 9). In melanoma, they act as adjuvants initiating the immune response and inducing effector activities redirecting cytotoxic CD8+ T cells against the cancer. DC vaccine response rates are still low, ranging from 10% to 20%, but they are the only immunotherapy so far that increase patient survival rates 53.
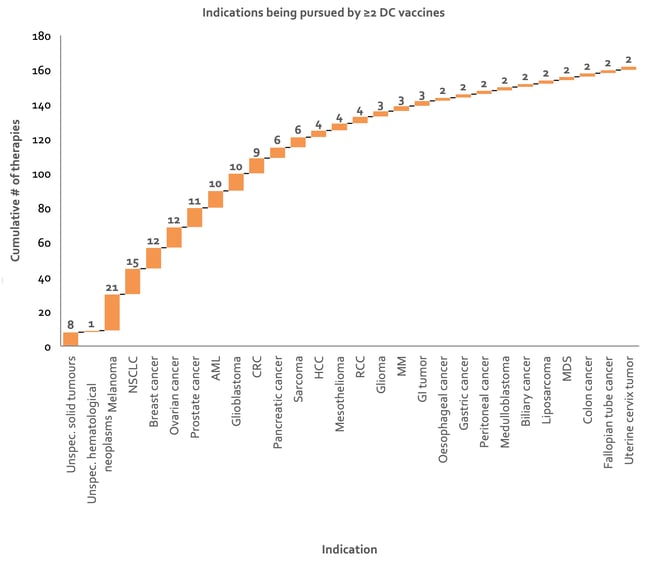
Figure 9: Waterfall chart depicting indications being targeted by at least two DC vaccines. Approximately 17% of the assets represented here are under collaboration, therefore resulting in some duplication of counts of number of therapies (data from Clarivate’s Cortellis - accessed H1 2020). AML, acute myeloid leukemia; CRC, colorectal cancer; HCC, hepatocellular carcinoma; MM, multiple myeloma; NSCLC, non-small cell lung carcinoma; RCC, renal cell carcinoma; MDS, myelodysplastic syndrome; GI, gastrointestinal.
γδ-T Cell, CIK and Macrophage Therapies
Pipeline γδ T-cell therapies are being developed to treat a range of cancers: ALL; gastric cancer; GBM; RCC, melanoma; NSCLC; myelodysplastic syndrome, AML, chronic myelogenous leukemia (CML), lymphoma, nasopharyngeal carcinoma, sarcoma, breast cancer, colorectal cancer (CRC) and prostate cancer. All CIK therapies in development are targeting solid tumor indications: esophageal cancer; gastric cancer; ovarian cancer; NSCLC; gastric cancer and hepatocellular carcinoma. The three CAR-macrophage therapies being developed by Carisma Therapeutics are targeting unspecified solid tumors. The indications being targeted by the macrophage therapies developed by Jounce Therapeutics Inc are also currently undisclosed.
Delivery methods
While cellular immunotherapies can be delivered via many different routes, the most popular by far is intravenous (IV) infusion, with 571 therapies being developed to be administered via this route. Its popularity is mainly due to convenient and effective drug administration that provides the most complete drug availability in a short period of time 65. The exception is for DC vaccines, which are most often delivered intradermally (ID). Vaccines are actually rarely given ID; however, research suggests that this route may be superior to the conventional subcutaneous (SC) or intramuscular (IM) route of vaccination 66. Some studies suggest that ID vaccination results in superior anti-tumor T cell induction compared with other routes of administration, such as intranodally 67. This might explain why the majority of DC vaccines are administered ID.
Certain other delivery methods improve the efficacy and safety of therapies. The TRAP method refers to an intraductal microcatheter trans-papillary technology for delivery of CAR-T cells into the breast ducts. This technology helps to reduce therapy toxicity 54. The benefit of intra-tumoral delivery is the increased amount of therapy directly reaching the target site and the reduced risk of systemic toxicity 55. On such example is Shanghai GeneChem’s anti-GPC3 targeting CAR that can be delivered intratumorally 56.
Studies have also found that intraperitoneal (IP) delivery of CAR-Ts is better at targeting peritoneal tumors than systemically infused CAR-Ts 57. The same has been found for intracerebroventricular (ICV) delivery of CAR-T therapies targeting central nervous system (CNS) tumors 58 and hepatic arterial infusion of CAR-T therapies for liver tumors 59. This is likely because locoregional delivery of the therapy allows the T cells to directly interact with the tumor cells and circumvent anatomical barriers involved in trafficking the T cells through the circulation 58.
Combination Treatments
Cancer therapies are often more effective when given in combination since each therapy can act synergistically or additively. Combining therapies also reduces the likelihood of developing drug resistance as each therapy often works by different mechanisms, and the risk of adverse effects is also lessened given each therapy can be used at a lower dose without compromising efficacy 60. Currently, there are over 250 cellular immunotherapy combination trials listed on clinicaltrials.gov (see Table 1).
Table 1: Number of combination therapy trials of each type of cellular immunotherapy (clinicaltrials.gov).
|
Technology type |
# of combination trials |
|
DC Vaccine |
80 |
|
CAR-T |
64 |
|
CIK |
46 |
|
TIL |
32 |
|
TCR |
16 |
|
γδ-T cell |
10 |
|
CAR-NK |
3 |
|
Macrophage |
0 |
Combination with other Cancer Treatments
Chemotherapy
Studies have found that combining cellular immunotherapies with chemotherapies appears to have an additive or synergistic effect 61. Some combinations in development include Sorrento Therapeutics’ phase 3 trial of an anti-CEA CAR-T therapy combined with gemcitabine, paclitaxel and capecitabine 62, and Sun Yat-sen University’s phase 2 trial of anti-PD-1 antibody-activated TILs combined with XELOX (capecitabine plus oxaliplatin) 63. Chemotherapies treat cancers by inducing immunogenic cell death or by preventing tumors from evading immune system detection 64. But they can also enhance the efficacy of cellular immunotherapies in a number of ways. First of all, they can increase the sensitivity of tumor cells to the immunotherapy, for example, it has been shown that the tumor-cell surface receptor, mannose-6-phosphate (M6P) is upregulated by certain chemotherapies, thereby increasing the permeability of granzyme B (released by CTL) to tumor cells 65. Secondly, some chemotherapies such as docetaxel, paclitaxel and vinblastine promote tumor cell recognition through increasing calreticulin exposure, which in turn promotes tumor cell death. The dying tumor cells then release many tumor antigens for recognition 66. Thirdly, chemotherapeutic drugs improve tumor antigen presentation, as autophagy of the tumor cells promotes release of ATP which in turn increases the number of DCs and T lymphocytes that are recruited to the tumor cell bed for antigen presentation 67. Finally, chemotherapies like doxorubicin, fluorouracil, gemcitabine, and cyclophosphamide are able to inhibit immunosuppressive cells like regulatory T cells and myeloid-derived suppressor cells – this enhances the activity of the cellular immunotherapy 68.
Radiotherapy
Combining cellular immunotherapy with radiotherapy also synergistically enhances treatment efficacy 69 70. As such, the University of Florida is running a phase 1 trial of its total tumor RNA (TTRNA) pulsed DC cell vaccine in combination with focal radiotherapy 71. The Sheba Medical Centre is also conducting a phase 2 trial of TIL therapy following lymphodepletion and total body radiation (TBR) 72. It is thought that radiotherapy prevents tumor escape by unmasking the tumor to the immune system so that it can be detected again 73, therefore playing an important role in improving antigen presentation. Like with chemotherapy, radiotherapy promotes calreticulin, which promotes cell death 74. Furthermore, radiation downregulates CD47, which is an anti-phagocytosis protein signaller 75. Both of these factors prime immune cells for action, so any cells forming an immunotherapy would also be primed, thereby improving their efficacy. Radiation also upregulates MHC-1 expression (which is downregulated in tumor cells) in order to improve recognition of cancer cells by CTLs 76. Lastly, DNA damage caused by radiation may also create neoantigens for recognition by the immune system 77.
Stem Cell Transplantation
After radiotherapy or chemotherapy, allogeneic or autologous hematopoietic stem cell transplantation (HSCT) is sometimes used to replace peripheral blood or bone marrow stem cells. The cells are essentially new immune cells that are able to recognize non-self-tumor cells and destroy them - known as the ‘graft-versus-tumor’ (GVT) effect 78. However, this approach is used with caution as relapse is common after HSCT, which is sometimes followed by GvHD, immunosuppression and development of opportunistic infections. Though it seems that combining HSCT with cellular immunotherapies may help to treat and prevent relapse of hematological malignancies by targeting minimal residual disease in patients undergoing allogeneic HSCT 79. Studies have suggested that the period of lymphopenia following HSCT may be an opportune time to use an immunotherapy treatment approach, helping to combat the negative effects of HSCT by enhancing the anti-tumor effect and boosting the immune system 80. In turn, HSCT may augment the effects of cellular immunotherapy by enhancing engraftment of the cellular immunotherapy and allowing the cells to activate and expand 81. Examples of this approach include completion of a phase 2 trial investigating DC vaccines after HSCT 82 by the University of Michigan Rogel Cancer Center, and Incysus Therapeutics’ phase 1 trial of Expanded/Activated γδ (EAGD) T cell therapy following HSCT 83.
While most cellular immunotherapies are being investigated post-HSCT, both CAR-T and CAR-NK therapies are mostly being investigated for their effects prior to HSCT. This may have benefits in relapsed patients who have failed to achieve remission and require a bridging-therapy before receiving an allogeneic HSCT, possibly allowing them to achieve the required second complete hematologic remission (CR) in order to be considered ideal for HSCT [84]. Since CAR-T therapy shows much higher rates of CR for hematological neoplasms than chemotherapy (and CAR-NK therapies have also shown very promising results in the clinic), these CAR therapies may be useful in aiding patients to achieve CR prior to receiving HSCT 85.
Immune Checkpoint Inhibitors
CPIs prevent cancer cells from using immune checkpoints to evade the immune system. Currently approved CPIs target CTL antigen 4 (CTLA4) (Ipilimumab, Yervoy), PD-1 (Pembrolizumab, Keytruda; Nivolumab, Opdivo; Cemiplimab, Libtayo) and PD-L1 (Atezolizumab, Tecentriq; Avelumab, Bavencio; Durvalumab, Imfinzi) 86. Cellular immunotherapies are readily combined with CPIs in clinical trials for example, the University of Pennsylvania is currently running a phase 1 trial investigating EGFRvIII T cells in combination with pembrolizumab in a subset of GBM patients 87 and Iovance Biotherapeutics is conducting a phase 2 trial investigating lifileucel/LN-145 TIL therapy in combination with pembrolizumab 88.
These two types of treatment offer a synergistic benefit when combined. CPIs can increase the tumor microenvironment’s susceptibility to lymphocyte infiltration and thus increase the sensitivity of tumor cells to cytotoxicity. For example, the PD-1/PD-L pathway inhibits T cell effector functions, NK cells and induces T cell exhaustion within the tumor microenvironment – a problem encountered by many different cellular immunotherapies 89. Blockade of this pathway thus allows cellular immunotherapies to target the tumor better. This is well demonstrated in a study in which treatment with pembrolizumab helped boost the immune system, leading to re-expansion of CAR-T cell populations in patients that had received CAR-T therapy but had subsequently relapsed 90.
Immunotherapy Combinations
Multi-CAR-T Therapy Combinations
Some trials have shown that combining multiple CAR-T therapies together is an effective cancer treatment because it prevents tumor escape via loss of antigens - even if the tumor drops one antigen, the other CAR-T therapy will still be able to target a different antigen 91. In one example, a phase 2 single-arm trial in China of humanized anti-CD19 and anti-BCMA CAR-T cells in relapsed/refractory multiple myeloma resulted in an impressive 95% response rate and 43% CR 92.
CAR-T Therapy and DC Vaccines
There are currently two ongoing clinical trials (as per clinicaltrials.gov) testing the combination of a CAR-T therapy and a DC vaccine 93.The rationale behind this combination hinges on the ability of CAR-T cells to shrink or fully kill the tumor and the DC vaccine to eliminate any minimal residual tumor cells or cancer stem cells that either still remain, or those that may develop for reasons such as tolerance to the CAR-T cells or simply due to a lack of T cell persistence. So it seems DC vaccines may help to enhance CAR-T cell therapy efficacy by expanding T cell clonal populations and enhancing T cell activation and persistence 94; essentially preventing any remaining tumor cells from causing disease relapse.
CIK Therapy and DC Vaccines
The combination of CIK and DC vaccines is thought to increase anti-tumor toxicity 95 96 97. One study found that the combination significantly reduced Tregs (which promote tumor development by suppressing the immune system) as per decreased serological liver tumor markers such as AFP, CA199 and CA242 and gastric tumor markers like CA724. Furthermore, the tumor progression indicator, myeloid-derived suppressor cell (MDSC) count also decreased 98.
γδ-T Cell Therapy and DC-CIK Cells
Jinan University Guangzhou is conducting three phase 3 trials combining γδ-T cell therapy with DC-CIK cells 99–101. It is believed the combination may enhance therapeutic efficacy by combining a non-MHC restricted immune cell (γδ-T cells) with tumor-specific T cells (the DC-CIK cells), hence increasing the immune response to cancer cells. A study by the National University of Singapore investigated co-expansion of CIK cells with γδ-T cells in order for them to be used for CAR-T cell therapy. It was found that the expanded cells exhibited powerful antitumor cytotoxicity 102, which supports the use of this combination as a strong anti-cancer treatment.
DC Vaccines and TIL Therapy
The combination of DC vaccines and TIL therapies has been little explored. One study found that efficacy was lower using the combination vs. TIL therapy with lymphodepleting chemotherapy and IL-2 103. Another more recent phase 1 study found that the combination of TIL therapy and DC vaccination therapy showed a greater response rate compared to TIL therapy alone however, in this trial, patients underwent preconditioning and received IL-2 after dosing with TIL 104, somewhat impeding any clear conclusions. Further clinical trials will need to be conducted in order to assess any truly beneficial effects of this combination.
References
1 Z. Tao et al., ‘Cellular immunotherapy of cancer: An overview and future directions’, Immunotherapy. 2017, doi: 10.2217/imt-2016-0086.
2 J. Zhang and L. Wang, ‘The emerging world of TCR-T cell trials against cancer: A systematic review’, Technology in Cancer Research and Treatment. 2019, doi: 10.1177/1533033819831068.
3 J. Cheng et al., ‘Understanding the Mechanisms of Resistance to CAR T-Cell Therapy in Malignancies’, Frontiers in Oncology. 2019, doi: 10.3389/fonc.2019.01237.
4 E. L. Siegler, Y. Zhu, P. Wang, and L. Yang, ‘Off-the-Shelf CAR-NK Cells for Cancer Immunotherapy’, Cell Stem Cell. 2018, doi: 10.1016/j.stem.2018.07.007.
5 O. Nussbaumer and M. Koslowski, ‘The emerging role of γδ T cells in cancer immunotherapy’, Immuno-Oncology Technol., 2019, doi: 10.1016/j.iotech.2019.06.002.
6 A. J. Gentles et al., ‘The prognostic landscape of genes and infiltrating immune cells across human cancers’, Nat. Med., 2015, doi: 10.1038/nm.3909.
7 M. W. Rohaan, J. H. van den Berg, P. Kvistborg, and J. B. A. G. Haanen, ‘Adoptive transfer of tumor-infiltrating lymphocytes in melanoma: a viable treatment option’, J. Immunother. Cancer, 2018, doi: 10.1186/s40425-018-0391-1.
8 ‘First-Ever T-Cell Therapy for Solid Tumors shows Remarkable Results | MesoWatch’. https://mesowatch.com/first-ever-t-cell-therapy-for-solid-tumors-shows-remarkable-results/ (accessed Jun. 17, 2020).
9 ‘TIL Therapy Elicits Encouraging Activity in Advanced NSCLC | OncLive’. https://www.onclive.com/view/til-therapy-elicits-encouraging-activity-in-advanced-nsclc (accessed Jun. 17, 2020).
10 Y. Meng et al., ‘Cell-based immunotherapy with cytokine-induced killer (CIK) cells: From preparation and testing to clinical application’, Human Vaccines and Immunotherapeutics. 2017, doi: 10.1080/21645515.2017.1285987.
11 W. Zhang et al., ‘Chimeric antigen receptor macrophage therapy for breast tumors mediated by targeting the tumor extracellular matrix’, Br. J. Cancer, 2019, doi: 10.1038/s41416-019-0578-3.
12 W. W. Van Willigen, M. Bloemendal, W. R. Gerritsen, G. Schreibelt, I. J. M. De Vries, and K. F. Bol, ‘Dendritic cell cancer therapy: Vaccinating the right patient at the right time’, Frontiers in Immunology. 2018, doi: 10.3389/fimmu.2018.02265.
13 B. Mastelic-Gavillet, K. Balint, C. Boudousquie, P. O. Gannon, and L. E. Kandalaft, ‘Personalized dendritic cell vaccines-recent breakthroughs and encouraging clinical results’, Frontiers in Immunology. 2019, doi: 10.3389/fimmu.2019.00766.
14 ‘Technology | PDC*line Pharma’. https://www.pdc-line-pharma.com/technology/technology (accessed Jun. 17, 2020).
15 ‘Xconomy: For CAR-T Cancer Fighters in the Real World, Two Roads Diverge’. https://xconomy.com/national/2019/01/03/for-car-t-cancer-fighters-in-the-real-world-two-roads-diverge/ (accessed Jun. 17, 2020).
16 ‘Making CAR-T affordable and accessible | Laboratory News’. http://www.labnews.co.uk/article/2029805/making-car-t-affordable-and-accessible (accessed Jun. 17, 2020).
17 ‘Novartis still hasn’t solved its CAR-T manufacturing issues | BioPharma Dive’. https://www.biopharmadive.com/news/novartis-kymriah-car-t-manufacturing-difficulties-cell-viability/568830/ (accessed Jun. 17, 2020).
18 ‘CreaVax-RCC | JW CreaGene’. http://www.creagene.com/creagene/en/product/creavaxRCC.jsp (accessed Jun. 17, 2020).
19 ‘Table 5 Approved and marketed cancer vaccines’. https://www.nature.com/articles/nbt0209-129/tables/5 (accessed Jun. 17, 2020).
20 ‘Northwest Biotherapeutics DCVax ® Technology - Northwest Biotherapeutics’. https://nwbio.com/dcvax-technology/ (accessed Jun. 17, 2020).
21 ‘Northwest Biotherapeutics NW Bio Announces Two German Approvals: “Hospital Exemption” for Early Access Program with DCVax-L and Eligibility of DCVax-L for Reimbursement - Northwest Biotherapeutics’. https://nwbio.com/nw-bio-announces-two-german-approvals-hospital-exemption-for-early-access-program-with-dcvax-l-and-eligibility-of-dcvax-l-for-reimbursement/ (accessed Jun. 17, 2020).
22 ‘Northwest Bio gets $40M more for controversial brain cancer vaccine | FiercePharma’. https://www.fiercepharma.com/r-d/northwest-bio-gets-40m-more-for-controversial-brain-cancer-vaccine (accessed Jun. 17, 2020).
23 ‘Vaccine Branch | Center for Cancer Research - National Cancer Institute’. https://ccr.cancer.gov/vaccine-branch (accessed Jun. 17, 2020).
24 S. Ali et al., ‘The European Medicines Agency Review of Kymriah (Tisagenlecleucel) for the Treatment of Acute Lymphoblastic Leukemia and Diffuse Large B‐Cell Lymphoma’, Oncologist, 2020, doi: 10.1634/theoncologist.2019-0233.
25 ‘Pipeline - Innovative Cellular Therapeutics’. https://www.ictbio.com/pipeline/ (accessed Jun. 17, 2020).
26 ‘Technology :: Adaptimmune Therapeutics plc (ADAP)’. https://www.adaptimmune.com/technology (accessed Jun. 17, 2020).
27 ‘Medigene - Technologies’. https://www.medigene.com/technologies (accessed Jun. 17, 2020).
28 ‘NantKwest - Technology’. https://nantkwest.com/technology/ (accessed Jun. 17, 2020).
29 ‘DC-CIK In Combination With Chemotherapy ( Gio / Oxaliplatin or Cisplatin ) Versus First-line Chemotherapy for Locally Advanced Unresectable or Metastatic Gastric Adenocarcinoma Randomized Controlled Phase II Clinical Study of Treatment - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT02504229 (accessed Jun. 17, 2020).
30 X. P. Lan et al., ‘Immunotherapy of DC-CIK cells enhances the efficacy of chemotherapy for solid cancer: a meta-analysis of randomized controlled trials in Chinese patients’, J. Zhejiang Univ. Sci. B, 2015, doi: 10.1631/jzus.B1500003.
31 C. L. Chen et al., ‘Safety and activity of PD-1 blockade-activated DC-CIK cells in patients with advanced solid tumors’, Oncoimmunology, vol. 7, no. 4, Apr. 2018, doi: 10.1080/2162402X.2017.1417721.
32 X.-H. Chang et al., ‘Specific immune cell therapy against ovarian cancer in vivo and in vitro’, Ai Zheng, vol. 27, pp. 1244–1250, Dec. 2008.
33 ‘Smart molecules trigger white blood cells to become better cancer-eating machines -- ScienceDaily’. https://www.sciencedaily.com/releases/2017/09/170928142121.htm (accessed Jun. 17, 2020).
34 J. Wei, X. Han, J. Bo, and W. Han, ‘Target selection for CAR-T therapy’, Journal of Hematology and Oncology. 2019, doi: 10.1186/s13045-019-0758-x.
35 B. Liu, L. Yan, and M. Zhou, ‘Target selection of CAR T cell therapy in accordance with the TME for solid tumors.’, Am. J. Cancer Res., 2019.
36 M. Peng et al., ‘Neoantigen vaccine: An emerging tumor immunotherapy’, Molecular Cancer. 2019, doi: 10.1186/s12943-019-1055-6.
37 R. C. Abbott, R. S. Cross, and M. R. Jenkins, ‘Finding the keys to the CAR: Identifying novel target antigens for t cell redirection immunotherapies’, International Journal of Molecular Sciences. 2020, doi: 10.3390/ijms21020515.
38 B. C. Miller and M. V. Maus, ‘CD19-Targeted CAR T Cells: A New Tool in the Fight against B Cell Malignancies’, 2015, doi: 10.1159/000442170.
39 T. Ma, J. Shi, and H. Liu, ‘Chimeric antigen receptor T cell targeting B cell maturation antigen immunotherapy is promising for multiple myeloma’, Annals of Hematology, vol. 98, no. 4. Springer Verlag, pp. 813–822, Apr. 2019, doi: 10.1007/s00277-018-03592-9.
40 R. Thomas et al., ‘NY-ESO-1 based immunotherapy of cancer: Current perspectives’, Frontiers in Immunology. 2018, doi: 10.3389/fimmu.2018.00947.
41 E. Anassi and U. A. Ndefo, ‘Sipuleucel-T (Provenge) injection the first immunotherapy agent (Vaccine) for hormone-refractory prostate cancer’, P T, vol. 36, no. 4, pp. 197–202, Apr. 2011.
42 T. Jiang et al., ‘Tumor neoantigens: From basic research to clinical applications’, Journal of Hematology and Oncology. 2019, doi: 10.1186/s13045-019-0787-5.
43 ‘Donor blood cell-based platform – CytoMed Therapeutics Pte Ltd’. https://www.cytomed.sg/donor-blood-cell-based-platform/ (accessed Jun. 17, 2020).
44 ‘Pipeline’. https://www.adicetbio.com/pipeline/ (accessed Jun. 17, 2020).
45 M. Martinez and E. K. Moon, ‘CAR T cells for solid tumors: New strategies for finding, infiltrating, and surviving in the tumor microenvironment’, Front. Immunol., 2019, doi: 10.3389/fimmu.2019.00128.
46 K. Garber, ‘Driving T-cell immunotherapy to solid tumors’, Nat. Biotechnol., 2018, doi: 10.1038/nbt.4090.
47 T. N. Yamamoto, R. J. Kishton, and N. P. Restifo, ‘Developing neoantigen-targeted T cell–based treatments for solid tumors’, Nature Medicine. 2019, doi: 10.1038/s41591-019-0596-y.
48 X. Xu et al., ‘Mechanisms of Relapse After CD19 CAR T-Cell Therapy for Acute Lymphoblastic Leukemia and Its Prevention and Treatment Strategies’, Frontiers in Immunology. 2019, doi: 10.3389/fimmu.2019.02664.
49 ‘Acute lymphoblastic leukemia - NHS’. https://www.nhs.uk/conditions/acute-lymphoblastic-leukaemia/ (accessed Jun. 17, 2020).
50 S. Lee and K. Margolin, ‘Tumor-infiltrating lymphocytes in melanoma’, Curr. Oncol. Rep., 2012, doi: 10.1007/s11912-012-0257-5.
51 M. Lauss et al., ‘Mutational and putative neoantigen load predict clinical benefit of adoptive T cell therapy in melanoma’, Nat. Commun., 2017, doi: 10.1038/s41467-017-01460-0.
52 ‘Clinical Pipeline - Iovance Biotherapeutics’. https://www.iovance.com/clinical/pipeline/ (accessed Jun. 17, 2020).
53 C. Alvarez-Dominguez et al., ‘Dendritic cell therapy in melanoma’, Ann. Transl. Med., vol. 5, no. 19, p. 386, Oct. 2017, doi: 10.21037/atm.2017.06.13.
54 ‘Development Pipeline - Atossa Therapeutics’. https://atossatherapeutics.com/product-pipeline/ (accessed Jun. 17, 2020).
55 M. A. Aznar, N. Tinari, A. J. Rullán, A. R. Sánchez-Paulete, M. E. Rodriguez-Ruiz, and I. Melero, ‘Intratumoral Delivery of Immunotherapy—Act Locally, Think Globally’, J. Immunol., 2017, doi: 10.4049/jimmunol.1601145.
56 ‘A Study of GPC3 Redirected Autologous T Cells for Advanced HCC - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT02715362 (accessed Jun. 17, 2020).
57 S. C. Katz et al., ‘Regional CAR-T cell infusions for peritoneal carcinomatosis are superior to systemic delivery’, Cancer Gene Ther., 2016, doi: 10.1038/cgt.2016.14.
58 D. Akhavan, D. Alizadeh, D. Wang, M. R. Weist, J. K. Shepphird, and C. E. Brown, ‘CAR T cells for brain tumors: Lessons learned and road ahead’, Immunological Reviews. 2019, doi: 10.1111/imr.12773.
59 S. C. Katz et al., ‘Phase I hepatic immunotherapy for metastases study of intra-arterial chimeric antigen receptor-modified T-cell therapy for CEA+ liver metastases’, Clin. Cancer Res., 2015, doi: 10.1158/1078-0432.CCR-14-1421.
60 R. B. Mokhtari et al., ‘Combination therapy in combating cancer’, Oncotarget, vol. 8, no. 23. Impact Journals LLC, pp. 38022–38043, 2017, doi: 10.18632/oncotarget.16723.
61 J. Cui et al., ‘Combined cellular immunotherapy and chemotherapy improves clinical outcome in patients with gastric carcinoma’, Cytotherapy, 2015, doi: 10.1016/j.jcyt.2015.03.605.
62 ‘Study of Anti-CEA CAR-T + Chemotherapy VS Chemotherapy Alone in Patients With CEA+Pancreatic Cancer & Liver Metastases - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT04037241 (accessed Jun. 17, 2020).
63 ‘A Study of Combination of Anti-PD1 Antibody-activated TILs and Chemotherapy in Colorectal Cancer - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT03904537 (accessed Jun. 17, 2020).
64 L. A. Emens and G. Middleton, ‘The interplay of immunotherapy and chemotherapy: Harnessing potential synergies’, Cancer Immunol. Res., 2015, doi: 10.1158/2326-6066.CIR-15-0064.
65 R. Ramakrishnan et al., ‘Autophagy induced by conventional chemotherapy mediates tumor cell sensitivity to immunotherapy’, Cancer Res., 2012, doi: 10.1158/0008-5472.CAN-12-2236.
66 L. Senovilla et al., ‘An immunosurveillance mechanism controls cancer cell ploidy’, Science (80-. )., 2012, doi: 10.1126/science.1224922.
67 M. Michaud, X. Xie, J. M. B. S. Pedro, L. Zitvogel, E. White, and G. Kroemer, ‘An autophagy-dependent anticancer immune response determines the efficacy of melanoma chemotherapy’, Oncoimmunology, 2014, doi: 10.4161/21624011.2014.944047.
68 J. Xu, Y. Wang, J. Shi, J. Liu, Q. Li, and L. Chen, ‘Combination therapy: A feasibility strategy for car-t cell therapy in the treatment of solid tumors (review)’, Oncology Letters. 2018, doi: 10.3892/ol.2018.8946.
69 C. H. Son, G. F. Fleming, and J. W. Moroney, ‘Potential role of radiation therapy in augmenting the activity of immunotherapy for gynecologic cancers’, Cancer Management and Research. 2017, doi: 10.2147/CMAR.S116683.
70 R. Therapy and B. Cdks, ‘The Science Behind Radiation Therapy’, Am. Cancer Soc., 2014.
71 ‘Brain Stem Gliomas Treated With Adoptive Cellular Therapy During Focal Radiotherapy Recovery Alone or With Dose-intensified Temozolomide (Phase I) - No Study Results Posted - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/results/NCT03396575 (accessed Jun. 17, 2020).
72 ‘Adoptive Cell Therapy Following a Reduced Intensity, Non-myeloablative, Lymphodepleting Induction Regimen in Metastatic Ovarian - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT03412526 (accessed Jun. 17, 2020).
73 W. Jiang, C. K. Chan, I. L. Weissman, B. Y. S. Kim, and S. M. Hahn, ‘Immune Priming of the Tumor Microenvironment by Radiation’, Trends in Cancer. 2016, doi: 10.1016/j.trecan.2016.09.007.
74 H. Yuan et al., ‘Multivalent bi-specific nanobioconjugate engager for targeted cancer immunotherapy’, Nat. Nanotechnol., 2017, doi: 10.1038/nnano.2017.69.
75 D. W. Vermeer, W. C. Spanos, P. D. Vermeer, A. M. Bruns, K. M. Lee, and J. H. Lee, ‘Radiation-induced loss of cell surface CD47 enhances immune-mediated clearance of human papillomavirus-positive cancer’, Int. J. Cancer, 2013, doi: 10.1002/ijc.28015.
76 J. P. Flynn, M. H. O’Hara, and S. J. Gandhi, ‘Preclinical rationale for combining radiation therapy and immunotherapy beyond checkpoint inhibitors (i.e., CART)’, Translational Lung Cancer Research. 2017, doi: 10.21037/tlcr.2017.03.07.
77 G. Germano et al., ‘Inactivation of DNA repair triggers neoantigen generation and impairs tumor growth’, Nature, 2017, doi: 10.1038/nature24673.
78 M. Cirillo, P. Tan, M. Sturm, and C. Cole, ‘Cellular Immunotherapy for Hematologic Malignancies: Beyond Bone Marrow Transplantation’, Biology of Blood and Marrow Transplantation. 2018, doi: 10.1016/j.bbmt.2017.10.035.
79 H. J. Kolb, B. Simoes, and C. Schmid, ‘Cellular immunotherapy after allogeneic stem cell transplantation in hematologic malignancies’, Current Opinion in Oncology. 2004, doi: 10.1097/00001622-200403000-00015.
80 M. N. Bouchlaka, D. Redelman, and W. J. Murphy, ‘Immunotherapy following hematopoietic stem cell transplantation: Potential for synergistic effects’, Immunotherapy. 2010, doi: 10.2217/imt.10.20.
81 C. Wrzesinski et al., ‘Hematopoietic stem cells promote the expansion and function of adoptively transferred antitumor CD8+ T cells’, J. Clin. Invest., 2007, doi: 10.1172/JCI30414.
82 ‘A Pilot Study of Tumor Cell Vaccine for High-risk Solid Tumor Patients Following Stem Cell Transplantation - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT00405327 (accessed Jun. 17, 2020).
83 ‘Expanded/Activated Gamma Delta T-cell Infusion Following Hematopoietic Stem Cell Transplantation and Post-transplant Cyclophosphamide - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT03533816 (accessed Jun. 17, 2020).
84 T. Hahn et al., ‘The role of cytotoxic therapy with hematopoietic stem cell transplantation in the therapy of acute lymphoblastic leukemia in adults: An evidence-based review’, Biology of Blood and Marrow Transplantation. 2006, doi: 10.1016/j.bbmt.2005.10.018.
85 S. Wen et al., ‘CAR-T bridging to allo-HSCT as a treatment strategy for relapsed adult acute B-lymphoblastic leukemia: A case report’, BMC Cancer, 2018, doi: 10.1186/s12885-018-5037-7.
86 ‘Immune Checkpoint Inhibitors and Their Side Effects’. https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/immune-checkpoint-inhibitors.html (accessed Jun. 17, 2020).
87 ‘CART-EGFRvIII + Pembrolizumab in GBM - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT03726515 (accessed Jun. 17, 2020).
88 ‘Study of Autologous Tumor Infiltrating Lymphocytes in Patients With Solid Tumors - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT03645928 (accessed Jun. 17, 2020).
89 M. van Gulijk, F. Dammeijer, J. G. J. V. Aerts, and H. Vroman, ‘Combination Strategies to Optimize Efficacy of Dendritic Cell-Based Immunotherapy’, Frontiers in immunology. 2018, doi: 10.3389/fimmu.2018.02759.
90 E. A. Chong et al., ‘Sequential Anti-CD19 Directed Chimeric Antigen Receptor Modified T-Cell Therapy (CART19) and PD-1 Blockade with Pembrolizumab in Patients with Relapsed or Refractory B-Cell Non-Hodgkin Lymphomas’, Blood, 2018, doi: 10.1182/blood-2018-99-119502.
91 N. N. Shah, T. Maatman, P. Hari, and B. Johnson, ‘Multi targeted CAR-T cell therapies for B-cell malignancies’, Frontiers in Oncology. 2019, doi: 10.3389/fonc.2019.00146.
92 Z. Yan et al., ‘A combination of humanised anti-CD19 and anti-BCMA CAR T cells in patients with relapsed or refractory multiple myeloma: a single-arm, phase 2 trial’, Lancet Haematol., 2019, doi: 10.1016/S2352-3026(19)30115-2.
93 ‘CAR-T Cells Combined With Peptide Specific Dendritic Cell in Relapsed/Refractory Leukemia/MDS - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT03291444 (accessed Jun. 17, 2020).
94 M. Capelletti et al., ‘Potent Synergy between Combination of Chimeric Antigen Receptor (CAR) Therapy Targeting CD19 in Conjunction with Dendritic Cell (DC)/Tumor Fusion Vaccine in Hematological Malignancies.’, Biol. Blood Marrow Transplant., vol. 26, no. 3, pp. S42–S43, Mar. 2020, doi: 10.1016/j.bbmt.2019.12.110.
95 D. Gao et al., ‘Autologous tumor lysate-pulsed dendritic cell immunotherapy with cytokine-induced killer cells improves survival in gastric and colorectal cancer patients’, PLoS One, 2014, doi: 10.1371/journal.pone.0093886.
96 X. Wang and I. Rivière, ‘Manufacture of tumor- and virus-specific T lymphocytes for adoptive cell therapies’, Cancer Gene Therapy. 2015, doi: 10.1038/cgt.2014.81.
97 R. Zhong, J. Teng, B. Han, and H. Zhong, ‘Dendritic cells combining with cytokine-induced killer cells synergize chemotherapy in patients with late-stage non-small cell lung cancer’, Cancer Immunol. Immunother., 2011, doi: 10.1007/s00262-011-1060-0.
98 Y. Wang et al., ‘The combination of dendritic cells-cytotoxic T lymphocytes/cytokine-induced killer (DC-CTL/CIK) therapy exerts immune and clinical responses in patients with malignant tumors’, Exp. Hematol. Oncol., vol. 4, p. 32, 2015, doi: 10.1186/s40164-015-0027-9.
99 ‘Safety and Efficiency of γδ T Cell Against Non Small Lung Cancer ( Without EGFR Mutation) - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT02425748 (accessed Jun. 17, 2020).
100 ‘Safety and Efficiency of γδ T Cell Against Breast Cancer(Her-, er-, and pr-) - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT02418481 (accessed Jun. 17, 2020).
101 ‘Safety and Efficiency of γδ T Cell Against Hepatocellular Liver Cancer - Full Text View - ClinicalTrials.gov’. https://clinicaltrials.gov/ct2/show/NCT02425735 (accessed Jun. 17, 2020).
102 S. H. Du et al., ‘Co-expansion of cytokine-induced killer cells and Vγ9Vδ2 T cells for CAR T-cell therapy’, PLoS One, 2016, doi: 10.1371/journal.pone.0161820.
103 I. Poschke et al., ‘A phase I clinical trial combining dendritic cell vaccination with adoptive T cell transfer in patients with stage IV melanoma’, Cancer Immunol. Immunother., 2014, doi: 10.1007/s00262-014-1575-2.
104 stina L. wickström et al., ‘Abstract CT032: Adoptive T cell transfer combined with DC vaccination in patients with metastatic melanoma’, 2018, doi: 10.1158/1538-7445.am2018-ct032.

