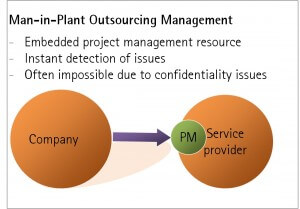
Many firms that outsource major projects can end up managing relationships at arms-length. For complex, business-critical projects, this is a high risk strategy as it may fail to give sufficient warning of project setbacks. Issues may only become actionable after they have become major problems. Embedding a “man in plant” is a good solution, but is often not practical due to the contractor’s cross-client confidentiality restrictions. In certain circumstances, a solution could be to integrate a trusted consultant in the contractor’s project team, allowing issues to be identified early and managed more effectively.
Biotech is accustomed to financial nuclear winters, but the novelty this time round is that the current funding crisis is not confined to biotech. It’s hitting the majors as well.
As ever, during cyclical downturns, the vogue is to make a virtue of necessity, and the mantra of the moment is ‘virtualize’. Outsourcing can underpin business efficiency and promote value creation – but only if it is implemented intelligently.
Virtual is not new
Biotech has always been virtual to one degree or other if for no other reason than economic efficiency. Biotechs seek external input in intellectual property, regulatory affairs, product development, market research and a whole host of other functions that may be considered ‘mission critical’, let alone those that are non-core. The same is true of the pharma, device and diagnostic majors, as evidenced by the large volumes of work placed by these companies with CROs.
Over recent years, the pendulum may have swung towards more outsourcing, but this has only really been an extension of a well-entrenched trend. The ongoing financial pressures, decreasing returns on R&D expenditures, an increasingly hostile reimbursement landscape and impending patent cliffs have simply exaggerated the need for operational efficiency. Sometimes, it just requires a simplification of overly complex internal structures and processes that have sclerosed the managerial arteries of the lumbering giants (hence the massive downsizing which shows no signs of abatement). Other situations, however, call for more virtualization/outsourcing, but getting this right requires high level expertise and experience and careful selection of the outsourcing partners.
Benefits of virtual mode are clear:
Human Resources
- Respond flexibly and cheaply to changing skills requirements
- Less bureaucracy (PAYE, pension, health insurance)
- For smaller companies: attract talent that would not otherwise join a startup
Cost effectiveness:
- A daily rate can sound expensive, but:
- Usually don’t need full time effort, so pay only for what you need
- Economic parity or better compared to fully-overheaded FTE
Lower risk:
- Try before you ‘buy’
- No severance risk/costs
- Fewer complications (salary scales, option grants)
Provider Selection: horses for courses
It may seem trivial to select the right type of partner for any given situation, but in reality these decisions are often taken without appropriate forethought. For example, it seems natural to turn to a CRO to manage a clinical trial, and indeed, for a large, multinational phase III trial this may be spot on. But for a small and/or specialized trial, the CROs’ tendency to force-fit the situation to their standard methodologies may be counterproductive. The typical CRO’s business model is driven by offering standardized services, usually with little true customization. Quality of service ranges to either extreme; one frustrated Big Pharma clinical researcher calls CROs “the people who work from 9 to 5 so we have to work from 8 to 10”.
It can be difficult for a large CRO to train their staff in ‘unusual’ techniques and procedures, not least because of their high staff turnover, and for non-standard trials it is usually better to engage a bespoke consortium of specialist service providers. In these situations, we have seen highly effective management of trials by companies who either engage relevant service providers directly or who contract with a specialist CRO with particular strengths in the disease/technology/trial design in question.
What to Outsource?
 Our firmly held belief, based on our team’s experience as consultants and senior managers in biotech and big companies, is that there is an optimal point between fully-integrated and fully-outsourced. A project or company needs a dedicated, full time core management team. They need enough skin in the game to keep them fighting in the trenches when challenges reach that critical point where those who are more detached would simply walk away. And in our view, one of the key responsibilities of the core management team is to decide which type of external input is needed for each specific situation.To many, the general rule of thumb is that anything that can be outsourced should be outsourced. It has been suggested that an entire management team can be composed of freelancers and consultants, i.e. taking the virtual concept to its extreme. We disagree with this proposition. It is a construct that is suited to endeavours where all goes
Our firmly held belief, based on our team’s experience as consultants and senior managers in biotech and big companies, is that there is an optimal point between fully-integrated and fully-outsourced. A project or company needs a dedicated, full time core management team. They need enough skin in the game to keep them fighting in the trenches when challenges reach that critical point where those who are more detached would simply walk away. And in our view, one of the key responsibilities of the core management team is to decide which type of external input is needed for each specific situation.To many, the general rule of thumb is that anything that can be outsourced should be outsourced. It has been suggested that an entire management team can be composed of freelancers and consultants, i.e. taking the virtual concept to its extreme. We disagree with this proposition. It is a construct that is suited to endeavours where all goes
well, but does not extend to the life sciences industry where, at the very least, biological variability of one sort or another conspires to derail programmes at multiple, often unexpected stages.
Effective Outsourcing Management
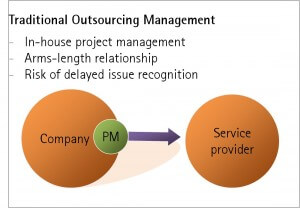
 Where large, business critical projects are outsourced, for example a clinical trial, there is scope for increased project risk due to the arms-length relationship between the parties.
Where large, business critical projects are outsourced, for example a clinical trial, there is scope for increased project risk due to the arms-length relationship between the parties.
The ideal solution would be to embed an employee in the contractor’s operations to provide maximum visibility on project progress and emerging issues. Unfortunately this is rarely an option given cross-client confidentiality concerns. This means that many customers manage their relationships with service providers at arms-length, risking delays in recognising emerging project roadblocks.
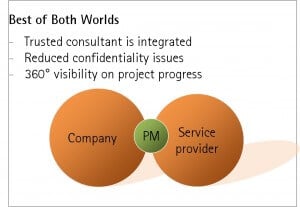
We propose a third way. Engaging with an experienced consultant to act as project manager presents a neutral interface for the contractor. This has benefits for companies carrying out virtual drug development including greater transparency into project progress, with a firewall to reduce confidentiality conflicts, as well as best-in-class project management at an appropriate cost.
Conclusion
The increasing virtualisation of operations has the potential to increase business risk due to the lack of visibility of project progress. Choosing an appropriate outsourcing strategy and taking an intelligent approach to project management can substantially mitigate this risk.
Download a PDF version:
Mitigating pharma biotech and medtech out-sourcing risk

